My own request to "please provide guidance" was answered by:
which I don't understand...
Now you may ask why I am so concerned about the addition of this new "derived" variable. Here are some issues:
- derived variables should not appear in SDTM. Again, the SDTM team has given in on a request from the FDA caused by primitive and immature review tools used by some FDA reviewers
- baseline flags should not appear in SDTM - they belong to ADaM
- sponsors should not be asked to do the work of FDA reviewers - the latter have to make their own decisions of which of the data points is "the" baseline data point.
- Assigning --LOBXFL is used to "camouflage" bad data quality. SDTM datasets with bad data quality should not be used in submissions and should not be accepted by the FDA.
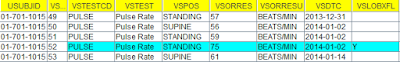
The following is a snapshot of a VS dataset with measurements done on the date "2014-01-02" which is also the date of first exposure (according to EX, and to RFXSTDTC in DM - another unnecessary derived variable) using the open source "Smart Dataset-XML Viewer":
According to the protocol, all vital signs measurements during this visit must be done before first drug intake. So the sponsor assigned the VSLOBXFL to the diastolic blood measurement with the value "76". What the sponsor however doesn't know, is that the researcher did the measurement immediately after the intake of the medication. As however too often, only the date (as well for the measurement as for the drug intake) was recorded, not the exact time.
Of course, the sponsor could also have assigned VSLOBXFL all three measurements on the date 2014-01-02, but as the standard does not specify "how" the derivation should be made ...
The same applies to the "PULSE" and "SYSBP" (systolic blood pressure) measurements:
If one inspects the data carefully, one will see that each of the "VSLOBXFL" records shows an increased value for that the specific measurent. This increased value may have been caused by the intake of the study drug. However, this is not visible, nor detectable, as no times have been collected (as is very usual) for either the measurement as the drug intake. Even worse, the increased value is marked as the baseline value, which may mean that the reviewer, when looking at later data points, comes to the conclusion that the drug is lowering blood pressure and pulse, whereas it is exactly the inverse...
How does the "Smart Dataset-XML Viewer" deal with such a situation?
One of the options of the "Smart Dataset-XML Viewer" is:
When however the measurement is on the same day as the first exposure, and either the time part of the measurement or of the first exposure is not provided, the "Smart Dataset XML Viewer" will highlight the record or records and provide a warning:
I pointed the SDTM team to all this in an additional review comment, which was answered as:
to which I responded:
Also the FDA reviewers have free access to the "Smart Dataset-XML Viewer", so they could use it too. On the other hand, the algorithm can also easily be implemented in SAS or any other modern review software.
As a conclusion, --LOBXFL is not only unnecessary, it also camouflages bad data quality. For reviewers, it is even potentially dangerous to trust on it as demonstrated above.
With --LOBXFL, it is just waiting for the first patient having his/her health seriously damaged ...








Thanks a lot for very important information about this controversial topic of --LOBXFL.
ReplyDelete